If you heat a pot of water on the stove ‘till it boils, what do you see? More to the point, what can you see through? The liquid water is clear, and the steam is clear. And you can tell the two apart.
Liquid water and steam (gaseous water) are two phases of the same substance. To boil water is to cause a phase transition because we start with one phase (liquid) and end up with another. There’s another special thing about this phase transition – there is both liquid and gaseous water in the pot at the same time (and again, we can clearly distinguish liquid water from gaseous steam). If we went in the opposite direction – cooling the steam to liquid – we’d see droplets of water condense bit by bit, just like we see bubbles of steam appear. Either way, the transition doesn't happen everywhere all at once. It happens in bits and pieces, and takes some time and a certain amount of energy (in the form of heat) before all the water has turned to gas.
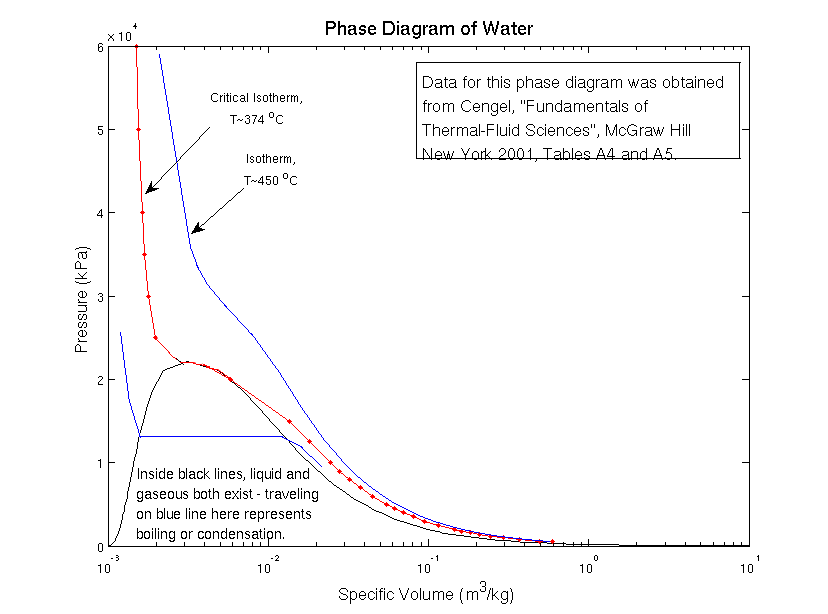 Look at the phase diagram above; it shows you at what temperatures and pressures water exists as a solid (ice), as a liquid, and as a gas (steam). A red line has been drawn to show the boiling transition on the phase diagram.
Look at the phase diagram above; it shows you at what temperatures and pressures water exists as a solid (ice), as a liquid, and as a gas (steam). A red line has been drawn to show the boiling transition on the phase diagram.

Ask yourself:
- What are some phase transitions you see all the time? What do they have in common?
- Why do we see the bubbles in the boiling water if liquid and gaseous water are both see-through?
- Click here for the answers!

Quizlet:
- Is an ice cube melting in a drink a phase transition?
- Is blending strawberries, bananas, and milk to make a smoothie a phase transition?
- Is burning a piece of wood a phase transition?
Click here for the answers!