The following experiment is described in a paper by E.S.R. Gopal of the Indian Institute of Science; you can download it for free (Resonance 5 (4), April 2000, 37-45).
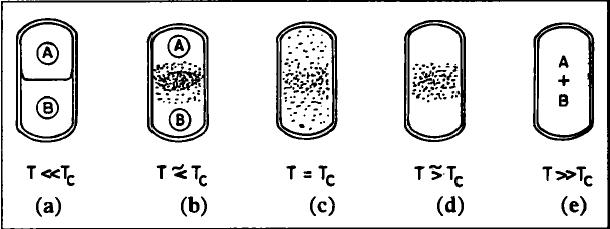
We said that the critical point of many common substances is beyond the temperatures and pressures of the human habitat. BUT that doesn’t mean that there aren’t systems with critical points at comfortable temperatures and pressures. One such system is the binary liquid (“two liquid”) combination of methyl alcohol, CH3OH, and carbon disulfide, CS2. Where before, we thought about the liquid, gas, and supercritical phases of the water example, now, we think in terms of two substances either being dissolved in one another or separate. Above the critical temperature, methyl alcohol dissolves in carbon disulfide – one single phase. Below it, the two substances separate into two distinct liquids – two phases. The same kind of opalescence happens at the critical point for the same reason.
Experiment:
External links:
A description of an experiment in critical opalescence for a binary fluid (different from one included in these pages):
- http://www.ucl.ac.uk/~uccaata/work/opalescence/opalescence.html
Description of binary liquid phase transition and critical point:
- http://www.msm.cam.ac.uk/doitpoms/tlplib/solid-solutions/demo.php