
Press Release: The 1987 Nobel Prize in Physiology or Medicine

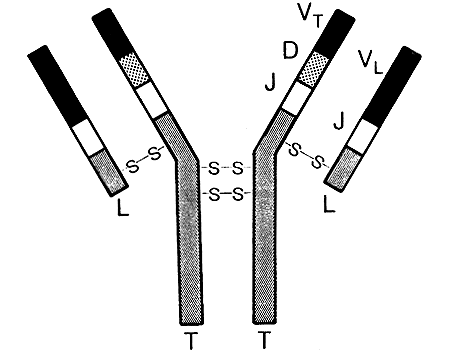
Figure 1. A picture of an antibody molecule with two long (T)
and two short (L) polypeptide chains which are kept together by sulphur
bridges (-S-S). The variable parts of a long chain (V,D, and J) and a light
chain (Y and J) together form the antigen binding area of the antibody.
In man there are five different types of long chains which have been given
letters M, D, G, A and E. The naming of the long chains forms the bases
for the names of the five so called immunoglobulin classes:
IgM, IgD, IgG and so on. The short chains are of two types: kappa or lambda.
Each antibody molecule has - regardless of class - either two kappa or two
lambda chains.
Towards the base of the Y there it is a constant part where the sequence
of amino acids is the same in all antibodies belonging to the same class.
In the outer ends of the two arms of the Y, however, there exist a significant
variation in the amino acid sequence when comparing different antibodies.
In this variable part there are three areas where variation is very
large. These areas constitute the walls in a "pocket" where the
foreign substance, the antigen, will fit and can bind. You can make the
analogy of an antibody molecule with a lobster where the claws of the lobster
correspond to the antigen binding parts of the antibody.
Through its Y-form the antibody accordingly is endow two identical antigen
binding areas. These areas have a more or less good fit to a particular
antigen. The better the fit the harder to grip of the antigen and the more
efficient the defense. As we are continuously confronted within an enormous
variety of antigens we also have to have a large number of molecules there
the variable parts do fit to different antigens.
The constant part of the antibody does also contain important biological
functions. After the binding of the antibody to an antigen on the surface
on for example a virus (Figure 2) the antibody molecule is changed in such
a way that its constant part will activate important parts of the immune
defense. Among these is the complement system which can directly make holes
in bacteria and other microorganism and which also attract white blood cells
such as macrophages ("big eaters") and granulocytes to the battle
ground.

Figure 2. A polio virus particle is attacked by four IgG antibodies.
Through this attack the infectious capacity of the virus is destroyed. It
is mainly through this mechanism that polio vaccine is functioning.
The richness of variety, an equation which didn't add up
Antibodies are produced by a special kind of white blood cells which are
called B lymphocytes and which in an adult human being amounts to one million
million cells (1012). As a single B lymphocyte only can produce
its own unique antibody the number of different antibodies in an individual
can theoretically not exceed that of the number of B lymphocytes.
The information how antibodies should be constructed lies in the genome
of the B lymphocytes. One hypothesis suggested, that in this genome there
exist one gene responsible for each type of polypeptide chain in
the antibody. But here the problem was that the immune defense contains
hundreds of thousands times more different antibody types than there are
total number of genes in the B cells. The equation simply didn't add up
and the hypothesis had to be abandoned. It was replaced by a second one
which explained the almost limitless capacity of variation in antibodies
as a result of some changes in the DNA of the B cell during the development
of the individual.
Susumu Tonegawa was the one who finally answered the question how
the gene material in B cells could suffice to create the structures of a
seemingly endless number of different antibodies. In 1976 he could in a
convincing and elegant manner show how different immunoglobulin genes which
were far apart in the embryonic cell in the B lymphocyte had been moved
in closer contact. Under development from the germ cells (the sperm and
egg cell) to an antibody producing B lymphocyte the genes forming the immunoglobulins
had accordingly been redistributed. In subsequent experiment Tonegawa could
clarify how different pieces of the genome were moved around, recombined
and even could be "lost" to finally give rise to the DNA which
is found in the mature B lymphocyte.
In the human the genes for the long chains are present on chromosome 14,
for the kappa chains on chromosome 2 and for the lambda chains on chromosome
22. Thanks to Tonegawa's pioneering work we now know how many immunoglobulin
genes there are in man, how they are put together and how they can give
rise to this high number of different antibodies.
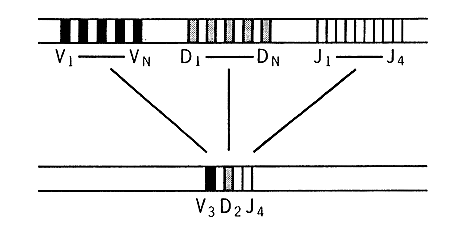
Figure 3. Redistribution of immunoglobulin genes for the long
chain during the development from an embryonic cell (top) to an antibody
producing B lymphocyte (bottom). Genes from each group V, D and J are brought
together in the final form for functioning gene for the variable part of
the long chain of an antibody molecule.
Economy through waste
Today we know that three groups of genes participate in the creation of
the variable part of the long chain, that is the part which together with
the variable part of the short chain is specific for each antibody. These
genes have the names V, D and J (Figure 3). The short chain has V and J
genes. In man the number of different Y genes for the long chains are around
200 to which should be added about 20 D genes and 4 J genes. When the functioning
gene of an antibody is to be created a single V, D and J gene are drawn
at random from the three groups of genes. The process can be compared to
a numbers lottery (Figure 4). 200 x 20 x 4 will give rise to 16 000 different
variable parts.

Figure 4. A registration sign for a car with its unique registration
number produced through lottery can illustrate the process which leads to
the creation of a unique antibody molecule. This registration number stands
for Susumu Tonegawa, the 144th Nobel Laureate in Physiology or Medicine.
V, D and J are put together in an irregular manner which will further enhance
the richness of variation. And as the V and D genes often are different
when inherited from our father and mother this will mean that already here
possibility has been created for something like five million different forms
of the variable part of the long chain. On top of this the light chain contributes
with more than 10 000 variants. The final sum will be many billions possibilities
of variation.
We are accordingly well prepared for an encounter with any possible antigen.
It is likely that normally only a minor part of the antibody variance will
ever be put into usage. The immune system is extremely economic when using
the DNA of the individual. At the same time a large number of lymphocytes
are produced and only a few of these will ever have to participate in the
immune defense of the body. The economy in the usage of DNA is thus combined
with a seeming waste of cells. This is, however, necessary to maintain the
high state of alertness which is required against possible new infections.
The discoveries of Tonegawa explain the genetic background allowing the
enormous richness of variation amongst antibodies. Beyond deeper knowledge
of the basic structure of the immune system these discoveries will have
importance in improving immunological therapy of different kinds, such as
for instance the enforcement of vaccinations and inhibition of reactions
during transplantation. Another area of importance are those diseases where
the immune defense of the individual now attack the bodies own tissues,
the so called autoimmune diseases.
References
P. Leder: The Genetics of Antibody Diversity. Scientific American 1982,
246, 72-83.
E. Norrby: Våra virus. Liber, Stockholm, 1987.
S. Tonegawa: Somatic Generation of Antibody Diversity. Nature 1983, 302,
575-581.
H. Wigzell: Vårt fantastiska immunförsvar. Liber, Stockholm,
1987.