ABSTRACT
King, J., Hall, C. and Casjens, S. (1978) Cell, 15(2), 551-560
Control of the synthesis of phage P22 scaffolding protein is coupled to capsid assembly
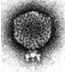
The assembly of the precursor shells of bacteriophage P22 entails the co-polymerization of gene 5 coat rotein with gene 8 scaffolding protein into double shell structures. During DNA encapsidation, the inner shell of scaffolding molecules dissociates and exits from the prohead. These molecules then recycle, catalyzing the assembly of newly synthesized coat proein to form new proheads (King and Casjens, 1974).
Although gene 5 and gene 8 are adjacent on the phage chromosome, we find that the synthesis of the two proteins is differentially regulated. In productively infected cells, scaffolding protein is synthesized at a low rate relative to the coat protein. In contrast, cells that are infected with mutants blocked in DNA packaging and accumulate precursor shells synthesize scaffolding protein at a much higher rate. If a mutation is introduced into the coat protein gene, however, preventing shell assembly, the rate of scaffolding protein systhesis decreases to less than the wild-type rate.
The experiments are consistent with models in which either continued synthesis of scaffolding protein depends upon co-polymerization with coat subunits, or soluble scaffolding subunits (but not assembled subunits) depress their own further synthesis. The finding that amber fragments of the scaffolding protein are synthesized at a very low rate is inconsistent with the second model. There is eidence, however, that fragments of the protein may have regulatory activity.
The regulatory circuit couples scaffolding protein synthesis to morphogenesis. Gene dosage experiments show that regulation results in the maintenance of coat and scaffolding subunits in the proper ratio for shell assembly.