Description
This page provides tables and a library of computational routines for the thermophysical properties of seawater. The properties given by these codes are those needed for design of thermal and membrane desalination processes. They are given as functions of temperature, pressure, and salinity.
The temperature and salinity ranges are 0 - 120 °C and 0 - 120 g/kg respectively. Some functions are valid only near atmospheric pressures while others are valid for a pressure range of 0 - 12 MPa. For thermodynamic functions, the pressure dependence across 0 - 12 MPa has been incorporated. For select functions such as transport and colligative properties, the temperature and salinity also implicitly define the pressure as:
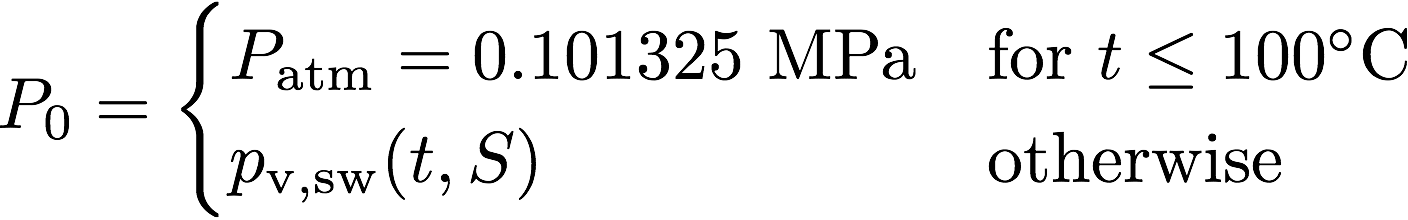
where P0 is taken as atmospheric pressure (0.101325 MPa) up to 100 °C and as the vapor pressure of seawater for higher temperatures.
Implementation and Fair Usage
The seawater properties library routines are available in MATLAB, EES, and Excel VBA. They are a self-contained library and are extremely easy to use. They will run on all computers that support MATLAB, EES, or VBA. This software is provided "as is" without warranty of any kind. See the file sw_copy.m or sw_copy.txt for conditions of use and license.
While the seawater properties library routines are freely distributed, it is requested that users of this library please cite the following two journal articles which comprehensively present how correlations in the library were developed, the original data sources, the validity ranges, and the uncertainty in each property function.
- K.G. Nayar, M.H. Sharqawy, L.D. Banchik, and J.H. Lienhard V, "Thermophysical properties of seawater: A review and new correlations that include pressure dependence," Desalination, Vol. 390, pp.1-24, 2016. doi:10.1016/j.desal.2016.02.024 (open-access preprint)
- Mostafa H. Sharqawy, John H. Lienhard V, and Syed M. Zubair, "Thermophysical properties of seawater: A review of existing correlations and data," Desalination and Water Treatment, Vol. 16, pp.354-380, April 2010. (PDF file of journal paper, including corrections through November 2023.)
Download Bibtex formatted references for these papers.
Downloads
- Tables of properties in pdf format (Version: 9 December 2023)
- MATLAB, EES, and VBA files in a zip archive (Version: 07 August 2024. Version history.)
Installation
For MATLAB:
- Place all the MATLAB m files in a directory called "Seawater"
- Add this folder to the MATLAB search path. See the MATLAB command "help path" for more details.
For EES:
- Place the files SEAWATER_EES.LIB and SEAWATER_EES.CHM in a directory called "Seawater"
- Place this folder inside the Userlib folder of EES. See the EES library routines for usage of the properties functions.
For Excel VBA:
- Place the files SEAWATER_VBA.xla in the default Excel AddIns folder.
- For Windows, default folder is typically "\Users\USER-NAME\AppData\Roaming\Microsoft\AddIns\"
Property Functions
| Property (unit) | Temperature, t (°C) | Salinity, S (g/kg) | Pressure, P (MPa) | Function name |
|---|---|---|---|---|
| Boiling point elevation (K) | 0 - 200 | 0 - 120 | P0 | SW_BPE(T, S) |
| Chemical potential of salts in seawater (J/kg) | 10 - 80 | 0.1 - 120 | 0 - 12 | ChemPot_S(T, S, P) |
| Chemical potential of water in seawater (J/kg) | 10 - 80 | 0 - 120 | 0 - 12 | ChemPot_W(T, S, P) |
| Density (kg/m3) | 0 - 180 | 0 - 150 | 0 - 12 | SW_Density(T, S, P) |
| Dynamic viscosity (kg/m-s) | 0 - 180 | 0 - 150 | P0 | SW_Viscosity(T, S) |
| Isobaric expansivity of seawater (1/K) | 10 - 180 | 0 - 150 | 0 - 12 | SW_IsobExp(T, S, P) |
| Isothermal compressibility of seawater (1/MPa) | 10 - 180 | 0 - 160 | 0 - 12 | SW_IsothComp(T, S, P) |
| Kinematic viscosity (m2/s) | 0 - 180 | 0 - 150 | P0 | SW_Kviscosity(T, S) |
| Latent heat of vaporization (J/kg) | 0 - 200 | 0 - 240 | P0 | SW_LatentHeat(T, S) |
| Osmotic coefficient | 0 - 200 | 0 - 120 | P0 | SW_OsmCoeff(T, S) |
| Osmotic pressure (MPa) | 0 - 200 | 0 - 120 | SW_OsmPress(T, S) | |
| Prandtl number | 0 - 180 | 0 - 150 | P0 | SW_Prandtl(T, S) |
| Specific enthalpy (J/kg) | 10 - 120 | 0 - 120 | 0 - 12 | SW_Enthalpy(T, S, P) |
| Specific entropy (J/kg-K) | 10 - 120 | 0 - 120 | 0 - 12 | SW_Entropy(T, S, P) |
| Specific flow exergy (J/kg) | 10 - 80 | 0 - 120 | 0 - 12 | SW_FlowExergy(T, S, P, Tds, Sds, Pds)* |
| Specific Gibbs energy (J/kg) | 10 - 120 | 0 - 120 | 0 - 12 | SW_Gibbs(T, S, P) |
| Specific heat capacity (J/kg-K) | 0 - 180 | 0 - 180 | 0 - 12 | SW_SpcHeat(T, S, P) |
| Specific internal energy (J/kg) | 10 - 120 | 0 - 120 | 0 - 12 | SW_IntEnergy(T, S, P) |
| Specific volume (m3/kg) | 0 - 180 | 0 - 150 | 0 - 12 | SW_Volume(T, S, P) |
| Surface tension (mN/m) | 0 - 100 | 0 - 131 | P0 | SW_SurfaceTension(T, S) |
| Thermal conductivity (W/m-K) | 0 - 180 | 0 - 160 | P0 | SW_Conductivity(T, S) |
| Thermal conductivity (pressure dep.) (W/m-K) | 10 - 90 | 0 - 120 | 0 - 12 | SW_ConductivityP(T, S, P) |
| Thermal diffusivity (m2/s) | 0 - 180 | 0 - 150 | P0 | SW_Diffusivity(T, S) |
| Vapor pressure (Pa) | 0 - 180 | 0 - 160 | SW_Psat(T, S) |
* Tds, Sds, and Pds are the dead state temperature, salinity, and pressure. The default values are Tds = 25 °C, Sds = 35 g/kg, and Pds = 0.101325 MPa
Seawater Surface Tension
The surface tension correlation used in this package is based on Nayar et al. (2014). They published reference data for the surface tension of seawater over the salinity range of 20 - 131 g/kg and a temperature range of 1 - 92 degC at atmospheric pressure. The uncertainty of the measurements varied from 0.18 to 0.37 mN/m with the average uncertainty being 0.22 mN/m. This data was correlated with an average absolute percentage deviation between measurements and the correlation of 0.19% and a maximum deviation of 0.60%.
- K.G. Nayar, D. Panchanathan, G.H. McKinley, and J.H. Lienhard V, "Surface tension of seawater," J. Phys. Chem. Ref. Data, 43(4):43103, Nov. 2014.
- doi:10.1063/1.4899037
- preprint:https://hdl.handle.net/1721.1/96884
- Bibtex formatted reference.