Research
The Bell Lab is studying the assembly of the multi-enzyme replisomes that replicate animal chromosomes and how these events are regulated during the cell cycle to ensure genome maintenance.
Chromosomes are the permanent repositories of the information that directs cell function. Each chromosome consists
of a single DNA molecule with an array of associated proteins. Each time a cell divides it must accurately and completely replicate
the DNA at the foundation of each chromosome and reassemble the proteins that interpret this essential cellular blueprint. Remarkably,
during an average human lifetime this process accurately synthesizes trillions of miles of DNA. Inaccurate, incomplete, or excessive DNA
replication can lead to cell death, developmental abnormalities, or cancer. Consistent with its critical role, chromosome duplication is
carefully coordinated with cell division.
Our studies focus on the events that occur at the starting points of chromosome duplication, called origins of DNA replication. These DNA sequences
are found at multiple sites on each eukaryotic chromosome and direct the assembly of a bidirectional pair of replication machines, or replisomes,
that replicate the DNA on both sides of the origin. Each replisome must coordinate the activity of multiple enzymes, including a DNA helicase,
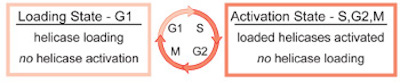
three DNA polymerases, and an RNA polymerase. The events that occur at origins can be broken down into four steps: origin selection, helicase loading,
helicase activation, and replisome assembly. To maintain proper chromosome numbers, dividing cells separate origin selection and helicase loading from
helicase activation and replisome assembly. This separation ensures that each origin of replication can initiate replication only once per cell division,
preventing rereplication of the genome (Fig. 1).
Figure 1
The six-protein origin recognition complex (ORC) mediates the initial selection of origin DNA sequences. ORC was
initially discovered in the budding yeast Saccharomyces cerevisiae, through its ability to bind to a conserved sequence
present in this organism's origins of DNA replication. Although these sites are present in many positions across the genome,
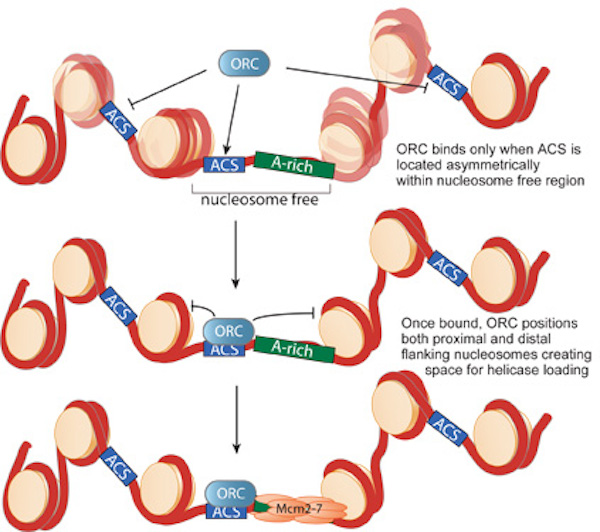
only a subset are bound by ORC. In collaboration with David MacAlpine's lab (Duke University), we used genome-wide mapping of
nucleosomes to demonstrate that nucleosome-depleted regions are a critical determinant of the sites bound by ORC (Fig. 2).
Once bound to origin DNA, ORC establishes the precise position of nucleosomes on both sides of the origin. Our previous experiments indicate that
this positioning is critical for helicase loading. We are currently focused on understanding how these nucleosome-depleted regions are
encoded at origins of replication and how the positioned nucleosomes influence helicase loading and replication initiation.
Figure 2
As cells enter the G1 phase of the cell cycle, ORC recruits two other helicase-loading factors (Cdc6 and Cdt1) and the replicative DNA helicase
(the Mcm2-7 complex) to the origin DNA. In a series of ATP-dependent events, ORC, Cdc6, and Cdt1 load
the DNA helicase onto the adjacent DNA. After loading, two ring-shaped, hexameric Mcm2-7 complexes encircle double-stranded DNA (dsDNA) as a head-to-head
double hexamer. Although inactive, these loaded double hexamers mark all potential origins of replication and are poised for bidirectional initiation.
We have investigated ATP control of helicase loading extensively. Analysis of Cdc6, ORC, and Mcm2-7 ATPase mutants reveals different roles for each
protein. Mcm2-7 ATP binding and hydrolysis are primarily responsible for the ATP requirement for helicase loading. Although Cdc6 ATP hydrolysis mutants
also contribute to helicase loading, this function is primarily involved in the release of Mcm2-7 complexes that fail to load. ORC ATP hydrolysis is not
required for helicase loading but is important for multiple rounds of Mcm2-7 loading and is essential in vivo.
Our latest studies use single-molecule approaches to address protein dynamics during helicase loading and provide insights into the mechanisms that
establish bidirectional replication initiation (Fig. 3). In collaboration with Jeff Gelles's laboratory at Brandeis University, we have developed
methods to monitor the association of multiple helicase-loading proteins with DNA by using colocalization single-molecule spectroscopy (CoSMoS). Our
studies reveal that the sequential loading of the two Mcm2-7 helicases in a double hexamer involves the ordered binding and release of distinct Cdc6
and Cdt1 molecules. In contrast, only one ORC molecule directs loading of both helicases in a double hexamer. Single-molecule fluorescence resonance
energy transfer (FRET) detection of Mcm-Mcm interactions shows that double hexamer formation occurs rapidly after arrival of the second helicase.
Together, our studies suggest that distinct mechanisms load the first and second helicase in each double hexamer and ensure that they are in the
head-to-head conformation required for subsequent bidirectional initiation. We are expanding these studies to monitor additional events during helicase
loading and to study mutant helicase-loading proteins to gain insights into the many new steps in helicase loading we have uncovered.
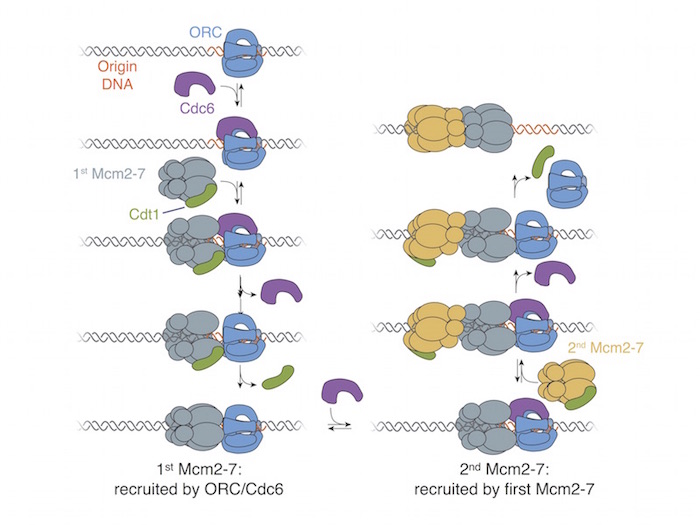
A model for the head-to-head loading of the Mcm2-7 double hexamer is shown in Figure 3. ORC recognizes origin
DNA and binds Cdc6. This complex recruits the Mcm2-7 helicase bound to Cdt1 by binding the C-terminal regions. Sequential release of Cdc6 and Cdt1
results in loading of the first Mcm2-7 helicase. After ORC binds a second Cdc6, the interactions between the N-terminal domains of the two Mcm2-7s
recruit the second helicase. Arrival of the second Mcm2-7 drives release of Cdc6, followed by Cdt1 and ORC. The result of this mechanism is loading
of the two helicases in opposite orientations that are poised for bidirectional replication initiation.
Figure 3
Modified from Ticau et al. (2015)
Mcm2-7 complexes loaded onto the origin DNA during G1 are only activated upon entry into the S phase of the cell cycle.
Helicase activation requires the action of the Dbf4-dependent Cdc7 kinase (DDK) and S-phase cyclin-dependent kinase (S-CDK).
These kinases trigger recruitment of the helicase-activating factors Cdc45 and GINS, forming the Cdc45-Mcm2-7-GINS (CMG) activated helicase.
In addition to activating the helicase, the cell must assemble the rest of the DNA replisome, including the three replicative DNA polymerases
that are dedicated to leading-strand (Pol ε) or lagging-strand (Pol α/primase, Pol δ) synthesis.
We used assays that recapitulate the origin-dependent initiation of replication to determine that DDK and S-CDK trigger distinct and sequential events
during helicase activation and that DDK is active prior to S phase at early origins of replication. By analyzing replisome assembly, we found that the
leading-strand DNA polymerase is recruited before its lagging-strand counterparts. This order of events ensures that the leading-strand DNA polymerase
is present before any RNA primer synthesis so that it can elongate the first primers.
We have used this replication assay to understand the functions of Mcm2-7 during replication initiation. Although many of the Mcm2-7 ATPase motif
mutations we studied were defective in helicase loading, a subset of these mutant complexes showed only minor defects. Analysis of these mutants
in the replication initiation assay revealed defects in CMG complex formation, suggesting roles for specific Mcm2-7 ATPase motifs in helicase activation.
We have also collaborated with Eric Enemark's lab (St. Jude Children's Research Hospital) to demonstrate that a newly identified single-stranded DNA–binding
domain in the N-terminal region of Mcm2-7 is also involved in helicase activation. Intriguingly, the polarity of this interaction predicts preferential
binding to the single-stranded DNA that the Mcm2-7 complex encircles during replisome movement. This observation suggests a mechanism for Mcm2-7 to
retain the translocating strand in the central channel during Mcm2-7's transition from encircling double-strand to encircling single-stranded DNA.
We continue to exploit these assays to gain insights into the mechanics of replication initiation and are pursuing single-molecule assays for these events.












