materials for cancer immunotherapy
Saturday, March 1, 2014
The immune system can actively attack tumors, but generally fails to control advanced cancers due to tolerance mechanisms (tumors “look like self” to the immune system) and immunosuppressive signals delivered in the tumor environment and possibly systemically in cancer patients. We are developing materials to deliver immunomodulatory factors or cells to tumor sites in order to promote the destruction of solid tumors and the generation of systemic immune responses against cancer that can eliminate metastases and protect the patient from recurrence.
One promising treatment for cancer in early stage clinical trials is known as Adoptive T-cell Therapy (ACT). In ACT, a patient’s own tumor-reactive immune cells (often, their T-cells) are isolated from tumor biopsies or peripheral blood, reactivated and expanded in cell culture, and then re-infused with the goal of having the patient’s own immune cells hunt down and destroy tumor cells throughout the body. To keep these transferred cells alive and functional, potent adjuvant drugs such as interleukin-2 are administered, which have serious dose-limiting toxicities that limit their use to only a subset of patients. Recently, we demonstrated a new strategy that both limits side effects from such adjuvant drugs while increasing their potency in promoting T-cell functions in vivo. In this approach, drug loaded nanoparticles are directly attached to the surface of live T-cells just prior to infusion (see micrograph above). The lymphocytes carry these drug-loaded particles with them wherever they traffic in the body, and receive continuous supporting signals as drugs leak out of the particles and bind to the cells’ surface receptors. Tiny doses of adjuvant drug can be used, thus limiting systemic side effects, while achieving very potent stimulation of the tumor-reactive T-cells. This is a general approach that can be applied to other drugs and other cell therapies, such as hematopoietic stem cell transplants.
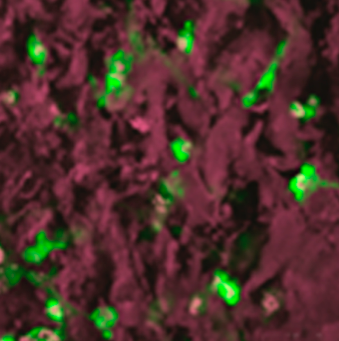
We have also developed biocompatible hydrogels (pictured purple fluorescence in above micrograph) that can be injected around solid tumors to release therapeutic cytokines or other factors in the tumor environment. This approach concentrates these factors in the tumor while lowering their concentration in the systemic circulation, compared to traditional i.v. injections. Immune cells are rapidly attracted to these matrices and subsequently infiltrate the neighboring tumor, leading to control of tumor growth. Other studies examining the injection of submicron-sized particles to release factors in the tumor environment and drive systemic immunity are also in progress.
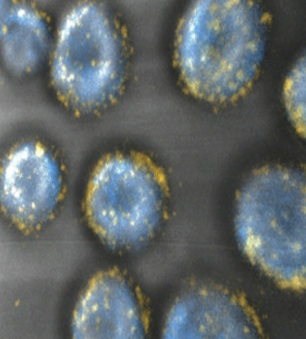
(Left) Injectable hydrogels (purple) are being explored to deliver immunomodulatory drugs to solid tumors. These synthetic polysaccharide gels are infiltrated by immune cells (green) that subsequently attack the neighboring tumor. (Right) Tumor-reactive “killer” T-cells (blue) decorated with drug-loaded nanoparticles (yellow) can be infused into tumor-bearing hosts, to hunt down and destroy tumors. By providing supporting drugs carried by each individual cell, the T-cells’ ability to proliferate and kill tumor cells is enhanced while minimizing exposure of other cell types to potent adjuvant drugs.