
July-September 1999 Issue
Capturing Carbon Dioxide
from Power Plants:
Cost-Effective Carbon Management?

July-September 1999 Issue
Capturing Carbon Dioxide
from Power Plants:
Cost-Effective Carbon Management?
![]() ne way to reduce emissions of greenhouse gases is to capture
and permanently store the carbon dioxide (CO2) emitted by electric
power plants burning fossil fuels. However, current methods of
capturing CO2 are expensive; so a critical concern with CO2 "capture
and sequestration" is cost. Energy Laboratory researchers have
performed a methodical study of projected costs for capturing CO2 from
three types of power plants, two fueled by coal and one by natural
gas. According to their analyses, capturing CO2 could push up the cost
of generating electricity from 3.3˘/kWh to 5.2˘/kWh at a natural gas
plant and from 4.6˘/kWh to 6.0˘/kWh at a coal plant based on
gasification. At those costs, carbon capture promises to be a
less-expensive near-term option for carbon mitigation than switching
to solar and perhaps nuclear power. With technological advances
expected by 2012, incorporating capture could add less than 1˘/kWh to
the cost of electricity. Increasing power plant efficiency could
reduce the cost of capture substantially as it would lower both the
cost per kWh generated and the amount of CO2 emissions to be captured.
Interestingly, if carbon capture and sequestration are practiced, coal
plants may become more competitive with natural gas plants as
restrictions on carbon emissions tighten. The researchers are now
using MIT's Integrated Global System Model to perform more rigorous
analyses of how CO2 capture compares to other carbon-mitigation
options under various assumptions about the future.
ne way to reduce emissions of greenhouse gases is to capture
and permanently store the carbon dioxide (CO2) emitted by electric
power plants burning fossil fuels. However, current methods of
capturing CO2 are expensive; so a critical concern with CO2 "capture
and sequestration" is cost. Energy Laboratory researchers have
performed a methodical study of projected costs for capturing CO2 from
three types of power plants, two fueled by coal and one by natural
gas. According to their analyses, capturing CO2 could push up the cost
of generating electricity from 3.3˘/kWh to 5.2˘/kWh at a natural gas
plant and from 4.6˘/kWh to 6.0˘/kWh at a coal plant based on
gasification. At those costs, carbon capture promises to be a
less-expensive near-term option for carbon mitigation than switching
to solar and perhaps nuclear power. With technological advances
expected by 2012, incorporating capture could add less than 1˘/kWh to
the cost of electricity. Increasing power plant efficiency could
reduce the cost of capture substantially as it would lower both the
cost per kWh generated and the amount of CO2 emissions to be captured.
Interestingly, if carbon capture and sequestration are practiced, coal
plants may become more competitive with natural gas plants as
restrictions on carbon emissions tighten. The researchers are now
using MIT's Integrated Global System Model to perform more rigorous
analyses of how CO2 capture compares to other carbon-mitigation
options under various assumptions about the future.
The world meets more than 85% of its energy needs using fossil fuels, and sufficient fuel supplies exist to continue that practice well into the 21st century. But there is a catch. Combustion of fossil fuels generates about 80% of all anthropogenic emissions of CO2, the single greatest contributor to potential global warming. Continuing our dependence on fossil fuels requires reducing those emissions, and an obvious target for change is the electric power industry. Within the United States, electric power plants generate fully a third of all anthropogenic CO2 emissions. In addition, power plants occur at limited locations so are more controllable than are other major sources of CO2 (transportation, space heating, and industrial processing). Strategies to reduce emissions from power plants include increasing fuel efficiency and developing non-fossil energy sources such as solar and nuclear power. But those strategies alone may not be sufficient, so international attention has also been focusing on a less-familiar approach: capturing the CO2 that comes out of fossil fuel-fired power plants and sequestering it, that is, recycling it or storing it to keep it out of the atmosphere. While a long-range strategy of decreasing dependence on fossil fuels is attractive, short-term major disruptions in our energy infrastructure have adverse economic consequences. Carbon capture and sequestration may be an effective transitional strategy.
For more than a decade, Energy Laboratory researchers led by Howard J. Herzog have been studying CO2 capture and sequestration. In early work they assessed the economic, technical, and environmental aspects of various methods of capture and sequestration (see e-lab, April-September 1989 and October-December 1992). One critical issue they identified is what to do with the huge quantities of captured CO2. Commercial uses for CO2 are limited, so the researchers have been examining methods of permanent storage, including injection into geological formations such as depleted oil and gas wells and into the deep ocean. Their analysis of the environmental impacts of deep ocean injection was described in the January-March 1996 issue of e-lab, and recent participation in ocean-injection field trials and other activities are discussed in the box at the end of this article.
Another issue the Energy Laboratory team has been addressing is cost. Some people believe that the cost of capturing and storing CO2 emissions would be exorbitant, largely due to the high cost of separating and capturing the CO2 prior to storage. Whether that conclusion is warranted is not obvious. For one thing, determining the cost of capture is tricky. Cost varies from one type of power plant to another and depends on many assumptions about plant design, performance, fuel use, and so on. Cost calculations are further complicated by the "energy penalty" associated with CO2 capture. The capture process uses energy, in some cases enough to reduce a plant's net output of electricity by 20%. Moreover, the energy used in the capture process creates additional CO2 emissions that must be captured. The decline in power output and the increase in CO2 emissions to be cleaned up must be reflected in the cost calculations. Finally, most cost estimates are based on today's CO2-capture technology, which was developed decades ago as a means of removing unwanted acid gases such as CO2 from process streams. The design and cost of a capture technique optimized for controlling power plant emissions may be quite different.
For the past year, Mr. Herzog and Neda Vukmirovic have been examining data from diverse sources to answer several key questions. First, what would the cost of electricity be if we incorporated CO2 capture capability into different kinds of power plants? Is there a clear winner in terms of plant design and fuel? How might costs change after a decade of research progress? And where are the greatest opportunities for making cost-reducing technological improvements?
The first task was to determine the cost of electricity and the levels of CO2 emissions for specific types of power plants, with and without CO2 controls. The researchers considered three types of CO2-capture plants.
Integrated Gasification Combined Cycle (IGCC): In this power plant, coal is gasified and reacted over a catalyst to form a mixture of CO2 and hydrogen. The CO2 is removed, and the remaining hydrogen powers a gas turbine combined cycle--an efficient source of electricity.
Pulverized Coal (PC): This conventional plant burns pulverized coal to raise steam, which drives an electricity-generating steam turbine. CO2 is removed from the exhaust gas using a scrubbing process involving the solvent monoethanolamine (MEA).
Natural Gas Combined Cycle (NGCC): In this plant, natural gas drives a gas turbine combined cycle, and CO2 is removed from the exhaust gases using the MEA scrubbing process.
The researchers used data from ten recently published studies that analyzed the economics of CO2 capture. All were based on commercially available equipment; all assumed effective controls on emissions of nitrogen oxides (NOx), sulfur oxides (SOx), and particulates; and all included the cost of compressing the captured CO2 for pipeline transportation. But critical assumptions such as fuel costs and discount rates differed from study to study. Therefore, the researchers developed a computer model that could adjust the various results so all were based on consistent economic assumptions. The model then synthesized the adjusted data from the individual studies to yield representative electricity cost and emissions data for each type of CO2-capture plant. For comparison, the model also calculated electricity cost and emissions data for conventional (non-capture) versions of the same types of plants based on data from the same ten studies and from the Coal Utilization Research Council (CURC).
Results from the researchers' analyses are shown in the figure below. The solid black symbols show the emissions level and electricity cost for each "reference" (non-capture) plant today. (Cost estimates are for generation only and exclude the cost of transmission and distribution. Today, a residential customer in the United States pays about 8˘/kWh for delivered electricity.) The hollow black symbols show what happens to those cost and emissions estimates when capture, separation, and compression are added to the plants.
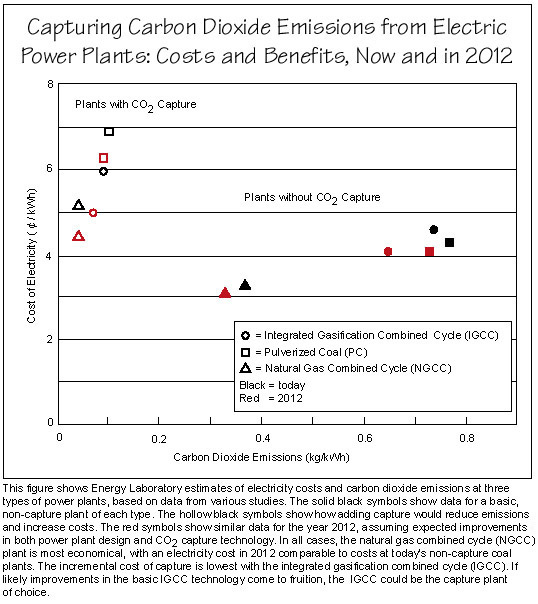
With today's technology, the NGCC performs best on both measures. The two coal plants emit about twice as much CO2 as the NGCC does; and the cost of electricity is higher, with the IGCC cost highest. With the CO2-capture process in place, emissions from all three plants drop substantially. Electricity still costs least at the NGCC plant. However, the cost of adding capture is lowest at the IGCC; and with capture installed, the IGCC plant becomes the more economical of the two coal plants.
Extensive research is now under way that promises to improve power plant designs and capture technologies. Therefore, the MIT investigators performed additional analyses assuming advances likely to be made by the year 2012. Based on information from the CURC, they assumed higher fuel efficiency and lower capital costs for the three basic plant designs. They also assumed improvements in today's capture technology, notably a decrease in energy required--a change that reduces both fuel costs and added emissions of CO2 due to the capture process.
The red symbols on the figure reflect results for the 2012 scenario. The solid red symbols show electricity cost and emissions for the three reference plants. The open red symbols show the effects of adding the optimized CO2 capture capability to the 2012 plants.
Technological improvements generally make the 2012 reference plants somewhat less expensive and cleaner than today's reference plants, with the IGCC technology making the greatest gains. Adding capture technology brings a similar reduction in CO2 emissions as in today's plants. But the cost of adding capture is lower in the 2012 plants. Even with the capture technology, the cost of electricity from the three types of plants is not exorbitant. In fact, the cost of electricity from the NGCC capture plant in 2012 is close to the cost of electricity from today's coal plants without capture technology.
Those results bring into question two attitudes that now prevail. First is the idea that CO2 capture and sequestration would be much more expensive than switching to non-fossil sources such as solar and nuclear power. While the MIT research is still in progress, the findings thus far suggest that carbon capture and sequestration may be a less expensive way to lower CO2 emissions than switching to technologies based on renewable energy sources. In particular, most projected solar electricity costs are well above the costs cited here.
The second prevailing attitude is that building new power plants that use natural gas rather than coal is the economically and environmentally sound choice. The analyses reported above support that belief, but there are several factors that could make coal competitive. For example, the cost of including capture capability is lower with the IGCC than with the NGCC. If the IGCC reference plant became less expensive, the coal capture plant could become a less-expensive source of electricity than the natural gas capture plant is. Also, if the price of natural gas rises by 30% and the price of coal remains the same, the cost per kWh becomes about equal with the two capture technologies. Both of those changes are feasible. Current research on the IGCC technology could lead to significant decreases in capital costs, and the electric power sector's expanding use of natural gas rather than coal will drive up demand for natural gas and hence perhaps its price.
Working with members of the Joint Program on the Science and Policy of Global Change, Mr. Herzog and Sean D. Biggs are now incorporating data from the CO2 capture analyses into MIT's Integrated Global System Model. This climate-change model combines descriptions of processes including economic growth, technological change, emissions growth, climate chemistry and physics, and ecosystem biology (see e-lab, January-March 1997). Using the Integrated Model, the researchers will perform rigorous analyses of how possible technology advances, fuel prices, carbon restrictions, and other policies and conditions would affect the economic viability of the different CO2 capture plants. In addition, they will see how the performance of those plants compares to that of other strategies, including switching to solar and nuclear power.
Mr. Herzog and his colleagues are now involved in a variety of other studies relating to CO2 capture. For example, they are performing sensitivity studies to see how much the cost of capture would be affected by increasing the power plant's efficiency and by decreasing the energy consumed in the capture process. Results thus far show that research priority should be placed on achieving efficiency gains. Increasing efficiency decreases not only electricity cost but also CO2 emissions. With CO2 emissions reduced, subsequent efforts to capture and sequester CO2 are less costly. According to their analyses, increasing the efficiency of today's IGCC power plant by 50% would make it possible to capture CO2 and reduce electricity cost simultaneously.
In other work, Mr. Herzog and his coworkers are examining different technologies for capturing CO2; they are considering possible methods for integrating CO2 capture with controls for other emissions; and they are looking at opportunities for retrofitting existing plants for CO2 capture. They are also continuing their work on CO2-storage methods and are planning systems analyses of how to coordinate capture and separation with transportation and storage to minimize overall costs. Finally, they are looking beyond 2012 at innovative technologies including new types of power plants and power cycles that may bring even larger reductions in the cost of capturing and sequestering CO2.
Howard J. Herzog is a principal research engineer in the Energy Laboratory. Neda Vukmirovic is a master's degree candidate in the Department of Chemical Engineering. Sean D. Biggs is a master's degree candidate in MIT's Technology and Policy Program. This research was supported by the US Department of Energy. Further information can be found in references.
|
Energy Laboratory Researchers Investigate
Ocean Sequestration of Carbon Dioxide The largest potential sink for carbon dioxide (CO2) captured from electric power plants is the ocean. The world's oceans now absorb about a third of our CO2 emissions, and they have the capacity to absorb far more. But the transfer of CO2 to the ocean is slow. Developing a faster and more direct means of moving CO2 into the ocean could potentially prevent dangerous spikes in atmospheric concentrations. However, the physical, chemical, and environmental effects of such actions are not well understood. Energy Laboratory researchers are now taking part in two major programs that bring together experts from various organizations to develop that understanding. Howard J. Herzog, principal research engineer in the Energy Laboratory, is participating in the US Department of Energy's Center for Research on Ocean Carbon Sequestration, or "DOCS." DOCS will receive a total of $3 million over three years to perform research on the feasibility, effectiveness, and environmental acceptability of two methods of ocean carbon sequestration. One method involves injecting CO2 into the deep ocean; the other calls for fertilizing marine organisms living on the ocean's surface so that they absorb more CO2. Research will combine observations and experiments in the ocean with computer modeling of ocean currents and the diffusion of CO2. DOCS is led by a consortium of the Lawrence Berkeley and Lawrence Livermore National Laboratories. Other participants are Moss Landing Marine Labs, the Pacific International Center for High Technology Research, Rutgers University, and the Scripps Institution of Oceanography. DOE formed DOCS and another center focusing on sequestration of CO2 in terrestrial ecosystems during the summer of 1999 as part of its program of research on global climate change. In other work, Mr. Herzog and Dr. E. Eric Adams, senior research engineer and lecturer in MIT's Department of Civil and Environmental Engineering, are taking part in an international field experiment that involves injecting small amounts of pure liquid CO2 into the deep ocean. Data gathered in the vicinity of the CO2 injection point will improve the basic understanding of underlying physical phenomena and will increase the accuracy of predictive computer models that are needed to evaluate the environmental impacts of CO2 injection. The CO2-injection experiment will take place on the Kona coast of the island of Hawaii during the summer of 2001. About 40 hours of daytime testing will occur over a period of about two weeks. Liquid CO2 will be injected at different flow rates through a small steel pipeline following the sea floor to an injection depth of nearly 3,000 feet. The experiment is being managed by the Pacific International Center for High Technology Research. Mr. Herzog and Dr. Adams are members of a technical committee that is supervising the design of the experiment and is responsible for the data collection and analysis. Other technical committee members include scientists and engineers from the United States, Japan, Norway, Canada, and Australia. The experiment is being sponsored by government agencies from the United States, Japan, Norway, Canada, and Australia and one private organization, ABB Corporate Research. For further information, go to the CO2 Ocean Sequestration Field Experiment Web site at <http://www.CO2experiment.org/>. |