
December 1998
Climate policy regarding perfluorocarbons (PFCs) may have a significant influence on investment decisions in the production of primary aluminum. This work demonstrates an integrated analysis of the effectiveness and likely economic consequences of different climate policy options. In our study we first compare atmospheric observations to the available estimates of PFC emissions for the baseline years 1990 and 1995. We then present projections for regional emissions of PFCs from the aluminum industry using the MIT Emissions Projection and Policy Analysis model under different policy scenarios. Abatement costs for emissions of PFCs and CO2 are compared in the context of the Kyoto Protocol.
Contents
1. INTRODUCTION
2. SOURCES OF PFC EMISSIONS
3. BASE ANALYSES USING ECONOMIC MODELS
4. PFC EMISSIONS REDUCTION: A COST-EFFECTIVENESS ANALYSIS
5. FUTURE GREENHOUSE GAS EMISSIONS FROM THE PRIMARY ALUMINUM INDUSTRY
6. CONCLUSIONS
7. REFERENCES
Seventy percent of world primary aluminum production capacity is currently located in the Annex B nations. The main greenhouse gas emissions associated with primary aluminum production are CO2 and the perfluorocarbons CF4 and C2F6.[1] Emissions of CO2 occur throughout the entire process chain as fossil fuels are used to provide the thermal and electric energy for the mining and processing of the raw materials, the electrolytic reduction of alumina, and the casting of the metal. The source of electric energy used for the electrolytic reduction is the single most important factor influencing total CO2 emissions from primary aluminum production. The specific emissions of CO2 vary by a factor of five depending on whether coal or hydroelectricity is used as a source of power for the reduction cells. [1] By contrast, emissions of PFCs are strictly the result of electrolytic reduction. They are formed only during the so-called anode effect when the electrolyte becomes depleted in alumina (Al2O3).[2]
In view of the PFCs' atmospheric stability and their large specific radiative forcing relative to most other greenhouse gases, increased scientific focus on these compounds is warranted. CF4 and C2F6 have estimated atmospheric lifetimes of at least 50,000 and 10,000 years, respectively, making them some of the most long-lived atmospheric pollutants. [3] Under the Kyoto Protocol the radiative impact of a greenhouse gas is expressed relative to an equal mass of carbon dioxide, through its global warming potential (GWP). The GWP is an aggregate index that takes into account a compound's absorption spectrum, how this overlaps with the spectra of other constituents of the current atmosphere, and the rate at which the compound is removed from the atmosphere over an integration period (agreed in the Kyoto Conference of Parties to be 100 years). CF4 and C2F6 exhibit GWP values of respectively 6,500 and 9,200. [4] One kilogram (kg) of CF4 in the atmosphere would thus be expected to have the same radiative effect as 6.5 metric tons of CO2 over a 100-year time horizon. (Throughout this paper we will use metric measures: 106 g = 10 3 kg = 1 T = 10-3 kT = 10 -6 MT.)
Aluminum producers in Annex B countries face the possibility that they, along with other industries, may be called upon by governments to make cuts in their emissions. To prepare for such a future contingency it is important to evaluate both the scope of technical options for the abatement of greenhouse gases, and the costs of such measures. This assessment is the purpose of this study, which consists of four parts. First, we quantify different sources of atmospheric PFCs, in order to establish an atmospheric baseline for emissions from the primary aluminum industry. Second, we describe the MIT Emissions Prediction and Policy Analysis energy-economic model and its use in projecting the growth of gross national product and the demand for primary aluminum, which is used to drive much of the subsequent analysis in the paper. Third, we utilize engineering data on aluminum reduction technology, PFC emissions, and the costs of smelter modernization to analyze the potential for reducing emissions of PFCs and CO2 in a cost-effective manner. Finally, we examine the influence of different technological factors on the future levels of greenhouse gas emissions from the primary aluminum industry.
The only major country for which quantitative information on the use of PFCs is available is Japan. In its second national communication under the UNFCCC it was reported that in 700 tons of CF4 were produced in 1995, 450 tons of which were consumed domestically and the remainder exported. [9] For C2F6, the corresponding number is 300 tons, none of which was exported. The Japanese figures are not corrected for destruction of the compounds in the process of their use.
The latest U.S. Greenhouse Gas Inventory reports aggregate emissions of 1.2 million tons of carbon equivalents (MTCE) from semiconductor manufacture for 1995. [10] Currently lacking better information, we assume a plausible mix of 50% C2F6, 30% CF4 and 20% C3F8 which exhibits an average global warming potential of about 8,000. U.S. emissions are thus broken down into 150 tons of CF4, 300 tons of C2F6 and 100 tons of C3F8. Further, assuming that the U.S., Japan and the "rest of the world" (for which no data are available) each account for one third of the world semiconductor production, we can derive a rough estimate for worldwide emissions of PFCs from the global semiconductor industry. For this purpose we use a value of 10% for the net destruction rate of CF4 in plasma etching. We also assume that Japanese manufacturing practice is representative of the situation in the "rest of the world." We thus obtain the following estimates for the global emissions of PFCs from semiconductor manufacture in 1995: 1100 +/- 500 tons of CF4, 700 +/- 300 of C2F6 and 50 +/- 40 tons of C3F8.
Other PFCs of uncertain origin have been detected in the atmosphere (for references, see [12]). These include c-C4F8, whose apparent global emission rate is 1,000-2,000 T/yr. It is likely that c-C4F8 is generated either as a by-product in the manufacture of fluoropolymers, or as an end-product of their degradation in incinerators. The origin of about 3000 tons of different atmospheric perfluoromethylcycloalkanes such as C6F11CF3, C5F9CF3 and C6F10(CF3)2 is also unclear. They may have leaked from high-voltage components of certain radar systems. These compounds have also had very limited use as tracers in meteorological dispersion studies. For the estimate quoted above, the atmospheric budget was calculated assuming constant emissions over a 30-year period.
No new atmospheric measurements warrant a revision of the earlier estimate for global emissions of C2F6 of 2,000 +/- 200 T/yr, calculated in Harnisch et al. [14] As the semiconductor industry's emissions of this compound have increased to 700 +/- 200 T/yr, the amount attributable to Al production is now 1,300 +/- 300 T/yr corresponding to a global mean specific C2F6 emission of 0.066 +/- 0.015 kg/T in 1995 from aluminum smelters. Similar to CF4, this corresponds to a 30% reduction relative to the late 1980s.
The results for our estimates for PFC emissions in 1995 are listed in Table 1. For CF4 a weak natural source was also included. [6] Globally, out of approximately 35 MTCE of PFCs emitted in 1995, roughly 60% were associated with the production of primary aluminum.
Table 1.Estimated Emissions of Various PFCs: 1995
| Gas | Observed Emissions in 1995 (T/yr) | Sources (T/yr) | Carbon Equivalents (MT/yr) |
| CF4 | 10500+/- 1000 | Primary aluminum (9400 +/- 1000) Semiconductors: primary - plasma etching; secondary - degradation of other PFCs (1100 +/- 500); Natural sources: (~1) |
17 2 |
| C2F6 | 2000 +/- 400 | Primary aluminum (1300 +/- 300) Semiconductors: plasma etching (700 +/- 300) | 32 |
| C3F8 | Primary aluminum (100 +/- 50) Semiconductors: plasma etching; fire extinguishers: halon replacement; refrigerants: R-413a (600 +/- 400) |
0.2 1.2 | |
c-C4F8 |
1000 - 2000 | Unknown: fluoropolymer pyrolysis? (1500 +/- 500) | 4 |
| C6F14 | Solvent (2500 +/- 500) | 5 | |
| C5F9CF3 C6F11CF3 C6F10(CF3)2 | Unknown (100 +/- 50) | 0.2 | |
| Total | 35 |
EPPA's highly aggregated, top-down architecture provides a stylized representation of the macroeconomic characteristics of various regions including sectoral production structure, patterns of demand for primary factors and intermediate goods, and assumptions about future technical change. It permits investigation of international agreements to limit greenhouse gas emissions (e.g., the Kyoto Protocol) via inter-regional comparison of the effects of reduction targets on global trade patterns, emission tax revenues, sectoral output shares, and growth of gross domestic product (GDP). For example, Kyoto binds the Annex B nations to a timetable of quantitative restrictions based on their greenhouse gas emission levels in 1990. We implement this policy in the model by placing bounds at the level of the regions in Table 2 on the aggregate carbon content of fossil fuels used in 2010 and subsequent periods. The Protocol's impact can then by analyzed by looking at the difference between the values taken by macroeconomic variables in the unconstrained "reference" and constrained "policy" cases.
Table 2. EPPA Regions
| Annex B | Non-Annex B |
| USA: United States | BRA:Brazil |
| JPN: Japan | CHN:China |
| EEC:European Union: Austria, Belgium, Denmark, Finland, France, Germany, Greece, Ireland, Italy, Luxembourg, Netherlands, Portugal, Spain, Sweden, UK | DAE:Dynamic Asian Economies: Hong Kong, Philippines, Singapore, South Korea, Taiwan, and Thailand |
| OOE:Other OECD nations: Australia, Canada, New Zealand, the European Free-Trade Area (excluding Switzerland and Iceland), and Turkey | EEX: Energy-exporting developing countries: OPEC states as well as other nations exporting oil, gas, and coal |
| EET: Eastern European economies in transition: Bulgaria, Czechoslovakia, Hungary, Poland, Romania, and Yugoslavia | IND: India ROW: Rest of World |
| FSU:Former Soviet Union: Russia and Ukraine |
Table 3. EPPA Sectors
| Production | Consumption | ||
| Agriculture | AGRIC | Food and beverages | FOODBEV |
| Coal | COAL | Final demand for energy | ENERGY |
| Crude oil | OIL | Transport and communications | TRNSPCOMM |
| Natural gas | GAS | Non-essential commodities | OTHER |
| Refined oil | REFOIL | ||
| Electric Power | ELEC | ||
| Energy-intensive industries | ENERINT | ||
| Other industries | OTHERIND | ||

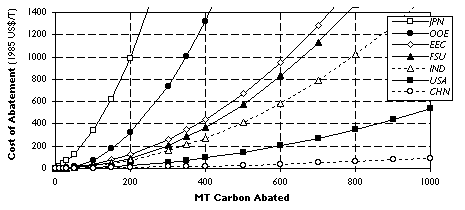
Figure 1. Marginal Abatement Curves for Carbon: Selected EPPA Regions in 2010.
The integral under each region's MAC up to the level of its emission reduction commitment measures that region's total abatement cost which is defined as the aggregate quantity of resources that the constraint on carbon makes unavailable for other uses in the economy at that point in time. MACs are a useful heuristic device because they allow easy determination of (1) the effect of a particular cut in emissions on the carbon price, and (2) the direct macroeconomic costs of a reduction policy in that period. For a detailed exposition of MACs and their use in CO2 emissions trading, see Ellerman and Decaux.[16]
PAD = a + b ln GDP
Data on country consumption of primary aluminum (in thousand metric tons) from 1960 to 1992 was taken from UNCTAD. [17], [18] Country time series of real GDP per capita (in constant 1985 US$) and population were extracted from the Penn World Tables Mark 5.6.[19] These data were aggregated to conform to EPPA's regional specifications before running the above regression. The results are shown in Table 4. To maintain consistency between EPPA's benchmark dataset and the data sources used in our econometric analysis, forecasts of GDP were constructed by scaling 1985 estimates of regional GDP from the Penn World Tables by GDP growth trends derived from runs of EPPA for the reference case. The resulting GDP forecasts and the parameters in Table 4 were then used to generate regional projections of the demand for primary aluminum to 2030 (Figure 2).
Table 4. Coefficients for regional Al Demand (thousand metric tons per year) as a Function of ln GDP (Billion 1985 US$)
| a | sa+ | b | sb+ | F | r2 | N | |
| USA | -18730.90 | (-6.51) * | 2812.90 | (7.85) * | 61.69* | 0.67 | 31 |
| JPN | -7182.49 | (-18.56) * | 1245.09 | (21.91) * | 480.24* | 0.94 | 31 |
| EEC | -24155.51 | (-31.17) * | 3383.75 | (34.57) * | 1194.88* | 0.97 | 31 |
| OOE | -7545.42 | (-21.39) * | 1308.87 | (24.29) * | 589.81* | 0.95 | 31 |
| EET | -1152.62 | (-9.51) * | 335.63 | (15.90) * | 252.74* | 0.89 | 30 |
| FSU | -9352.28 | (-13.52) * | 1561.87 | (16.01) * | 256.42* | 0.90 | 28 |
| EEX | -2729.48 | (-8.93) * | 436.27 | (9.84) * | 96.77* | 0.76 | 31 |
| CHN | -3185.03 | (-15.74) * | 551.59 | (18.05) * | 325.72* | 0.91 | 31 |
| IND | -1644.63 | (-19.07) * | 292.12 | (21.45) * | 459.97* | 0.94 | 31 |
| DAE | -1105.83 | (-7.10) * | 216.28 | (8.26) * | 68.28* | 0.72 | 26 |
| BRA | -1100.27 | (-14.94) * | 224.75 | (17.82) * | 317.57* | 0.91 | 31 |
| ROW | -2195.18 | (-12.40) * | 349.90 | (14.01) * | 196.42* | 0.86 | 31 |
| WORLD | -110256.98 | (-32.44) * | 12991.51 | (36.24) * | 1313.30* | 0.98 | 31 |
+ t-statistics, shown in parentheses.
| |||||||
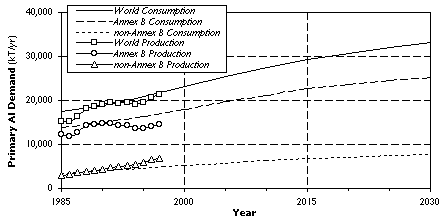
Figure 2. Projected Primary A1 Demand and Historical Production.
On the basis of these results we project a worldwide demand of 27.5 MT/yr for primary aluminum in 2010. Although the industrialized Annex B countries will most likely be responsible for the bulk of additional demand, we expect the rates of growth in demand to be much higher in the developing non-Annex B nations. In the late 1980s the Annex B countries have become net importers of primary aluminum, with consumption there continuing to grow while production has remained fairly constant. This aggregate behavior was the net result of new smelters being commissioned at the same time as old smelting capacity that was no longer profitable to operate was being scrapped. Throughout the same time-frame the non-Annex B countries expanded both their production and consumption of primary aluminum (to the point where they became net exporters in the late 1980s), a trend which is likely to continue well into the next century. At present it is unclear whether the Kyoto Protocol will accelerate or mitigate these global market trends. The outcome greatly depends on whether innovative financing mechanisms can be developed that help to abate greenhouse gas emissions in the Annex B countries, while at the same time preserving the competitiveness of their aluminum industry.
It must be remembered that in the case of PFC emissions from primary aluminum, conventional end-of-pipe abatement methods for air pollution (e.g., scrubbers) are generally unsuitable. This is due to PFCs' low concentrations in smelter exhaust gas streams, their poor aqueous solubility, and their high thermal and chemical stability. By contrast, recent studies seem to indicate that emissions of these compounds can be very efficiently abated by changes in process control and reduction technology. [2], [20], [21], [22], [23] Environmental benefits notwithstanding, the primary motivation for such technological changes is their effect on the economic performance of smelters, increasing power efficiency or labor productivity to reduce costs or expand output. It is therefore much more difficult to categorize specific components of expenditures on smelter modernization as "PFC abatement costs," in comparison with accounting for investments in air-pollution control technology. However, by abstracting from the site-specific benefits from technology conversion (which depend on factors such as energy prices or labor and capital costs) we can nonetheless derive rough estimates for the effect of changes in operating practices and technology on PFC emissions. Moreover, by relating these investments to their technical potential for PFC abatement, we are able to identify the extent to which they may be pursued as cost-effective options for reducing greenhouse gases.
An industry survey of the mean frequency and duration of anode effects at individual smelters [25] estimated that specific PFC emissions declined by 30% between 1990 and 1993. We used these data [[25]: p. 46, Fig. 10] with slight modifications to characterize the distribution of specific emissions for the four main smelter technologies above (see Figure 3) in a manner that ensures consistency with atmospheric observations.
For each technology the emissions distribution deviates substantially from the smooth S-shaped cumulative normal curve, indicating systematic intra-group differences among smelters. First, to capture the lower PFC generation achieved by modern Prebaked technology we created a new category "best available technology"-prebaked (BAT-PB), to which we assigned the smelting capacity with the lowest 30% of emissions under IPAI's CWPB classification. [25] We designated the capacity responsible for the remaining 70% of emissions as CWPB. Second, comparisons were made across a number of sources in the literature of estimates of mean specific PFC emissions (kg PFC/T Al) associated with the technologies above. [2], [21], [23], [25] In light of our estimates based on atmospheric observations we find that IPAI [25] overstates PFC emissions from VSS technology, where significant destruction of these compounds is likely to occur in the post-combustion of exhaust gases (Table 5). Due to a lack of data we derived preliminary specific emission estimates for C2F6 by assuming the CF4/C2F6 ratio of specific emissions to be 6 and 10 for Prebaked and Soderberg technologies, respectively. The resulting numbers were then scaled for consistency with the absolute emissions estimates of CF4 and C2F6 in Section 2.3, taking into account each technology's share of global smelting capacity. [25]
Table 5. Estimated Average Specific Emissions of CF4 / C2F6 from Different Smelter Technologies in 1993-1995 (kg/T aluminum)
| IPAI[25] | Leber et al.[23] | Bouzat et al.[21] | ||||
| CF4 | C2F6 | CF4 | C2F6 | CF4 | C2F6 | |
| CWPB | 0.3 | - | 0.10 - 0.25 | 0.02 - 0.06 | ||
| SWPB | 1.5 | - | 0.78 | 0.22 | ||
| VSS | 2.3 | - | 0.28 - 0.70 | 0.02 - 0.03 | ||
| HSS | 0.6 | - | 0.08 - 0.16 | 0.01 - 0.02 | ||
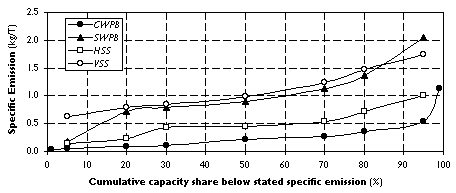
Figure 3. Distribution of Specific Emissions by Technology.
The results are shown in Table 6. These estimates are necessarily rough due to the assumptions that we have been forced to make in the process of their derivation, all of which are attributable to the general lack of data on this subject. In the future, sufficiently precise measurements of the temporal evolution of atmospheric mixing ratios of CF4 and C2F6 may make possible annual assessments of the progress of PFC emissions abatement in the aluminum industry.
Table 6. Breakdown of 1995 World Primary Aluminum Production by Smelter Technology, Assigned Specific Emissions, and Calculated Absolute Emissions
| World Production in 1995 (kT/yr) | Mean Specific Emission (kg/T) CF4 | Mean Specific Emission (kg/T) C2F6 | CF4 (T/yr) |
C2F6 (T/yr) | |
| BAT-PB | 3700 | 0.05 | 0.008 | 190 | 30 |
| CWPB | 7600 | 0.3 | 0.05 | 2270 | 380 |
| SWPB | 1900 | 1.2 | 0.02 | 2230 | 370 |
| VSS | 3700 | 1.1 | 0.11 | 4040 | 400 |
| HSS | 2000 | 0.4 | 0.04 | 780 | 80 |
| TOTAL | 18900 | 9510 | 1260 | ||
We assume that the potential for PFC abatement from improved plant operation was largely realized by 1995. For each technology category, estimates of conversion and retrofitting costs (in 1985 US$/T Al) were calculated from various sources in the literature and interviews with industry experts. [26], [27], [28], [29] We did not undertake a detailed capital budgeting analysis, but instead assumed that the cost of investments was amortized over 10 years at a 15% rate of discount to arrive at estimates for the annualized abatement cost per ton of primary aluminum. For conversion of the higher-emission smelter technologies to BAT-PB, the PFC reductions yielded by investment is the difference between the mean specific emissions of the existing and best available technologies given in Table 6.
The following procedure was used to compute the emission reductions from retrofitting. Because the distributions of specific smelter emissions in Figure 3 are neither uniformly nor normally distributed over the entire percentile range, we performed least-squares regression analyses on data-points in the linear regions of the cumulative distributions to calculate underlying trends in the shares of specific emissions for each technology. Based on these trends (shown in
Figure 4) we derived the mean reduction in specific emissions from retrofitting, assuming that in each technology group those 50% of smelters with emissions higher than median would benefit from investment. For each technology, we aggregated all non-retrofitted smelters in the upper 50th percentile into a single, representative data-point: the trend value of specific emissions corresponding to the 75% cumulative share. Similarly, we represented all retrofitted smelters by the trend value of specific emissions corresponding to the 25% cumulative share. Thus, for each technology, the reduction in specific emissions as a result of retrofitting is computed as the difference between the trend values associated with the 75% and 25% cumulative shares.
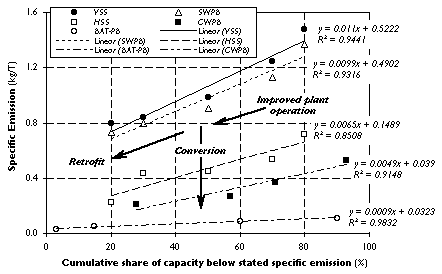
Figure 4. Trends in the Distribution of Specific Emissions.
Estimates of the specific costs of PFC abatement and quantity of emissions reduction that different options are likely to yield were used to construct a global average marginal abatement curve for PFCs, shown in Figure 5. The figure demonstrates that, depending on the climate policy scenario, different measures to reduce PFC emissions are likely to be cost-effective in order to comply with Kyoto. Within Annex B countries the extent to which greenhouse gas reductions in 2010 can be supplied at a given cost is likely to differ significantly across regions (Figure 1). The corollary is that each region's costs under the Protocol will depend strongly on the extent to which low- and high- marginal cost regions can engage in trading in emission rights. In a no-trading scenario where each Annex B region achieves its emissions reduction commitment under the Protocol through purely domestic measures, the shadow price (in 1985 US$) of carbon equivalents in 2010 is expected to vary from zero in FSU and EET to $180/ton in USA to as high as $500/ton in Japan. By contrast, these values fall to $100-150/ton with trade in emissions allowances within Annex B countries, and $20-30/ton when the full gains from emissions trade between Annex B and non-Annex B countries can be achieved. [16] Comparing these numbers with our results in Table 7 and Figure 5, in the no-trading scenario both the conversion and retrofitting of primary smelters would appear to be cost-effective tools of national climate policy in Annex B countries. In the event of intra-Annex B trading, such policies would probably only include retrofitting. With full global trade in emission rights, few additional PFC abatement measures would prove cost-effective, compared to the opportunities available for reducing greenhouse gas emissions from other economic activities.
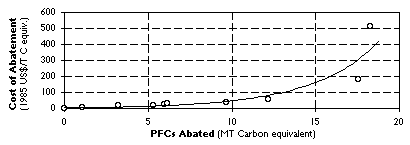
Figure 5. Marginal Abatement Curves for World A1 Industry PFC Emissions.
Table 7. Estimates of Cost and Technical Abatement Potential for PFCs
in 1995 (Investment, annual cost and CF4 reduction are given per installed ton of annual capacity)
| Measure |
Total Investment+ ($/T) | Annual Cost+ ($/T) | CF4 Reduction (kg/T) | PFC Reduction Cost+ ($/TCE) | Eligible Capacity Worldwide (MT) |
Total PFCs Abated (MTCE) | |
| BAT-PB | Improved control | 15 | 4 | 0.05 | 33 | 1.9 | 0.2 |
| CWPB | Conversion to BAT-PB | 150 | 36 | 0.28 | 59 | 7.6 | 4.6 |
| CWPB | Moderate retrofit | 40 | 9 | 0.25 | 17 | 3.8 | 2.1 |
| SWPB | Conversion to BAT-PB | 360 | 91 | 1.10 | 38 | 1.9 | 4.6 |
| SWPB | Moderate retrofit | 40 | 9 | 0.50 | 8 | 1.0 | 1.1 |
| VSS | Conversion to BAT-PB | 1500 | 364 | 1.00 | 180 | 3.7 | 7.5 |
| VSS | Moderate retrofit | 70 | 18 | 0.55 | 16 | 1.9 | 2.1 |
| HSS | Conversion to BAT-PB | 1500 | 364 | 0.35 | 514 | 2.0 | 1.4 |
| HSS | Moderate retrofit | 70 | 18 | 0.33 | 27 | 1.0 | 0.7 |
+ All costs in 1985 US$ | |||||||
A few caveats are worth mentioning here. First, because our analysis of retrofitting generally excludes the upper deciles of the emissions distributions, the slope of our calculated share trends will tend to be biased downwards. Our procedure for estimating specific abatement thus understates the potential quantity of emissions reduction, implying that our estimates of the potential abatement per dollar of investment may be conservative. Second, by comparing projected abatement in 2010 with currently available PFC reductions, the foregoing analysis implicitly assumes that reference PFC emissions will remain constant at 1995 levels. The major uncertainties in this regard are the industry's ability to introduce new low-emission smelting technology or to maintain historical rates of energy efficiency improvement. Factors such as these may well generate large additional reductions in both PFC emissions and production costs. A case in point is the approximately 9 MTCE of additional abatement worldwide achieved by the aluminum industry in 1990-1995 at low or negative cost, through improved plant operation. Finally, in the absence of a thorough site-by-site analysis of investments in capacity upgrading that takes into account both technological opportunities and economic conditions, the MAC shown in Figure 5 is necessarily illustrative. Nevertheless, in light of the sparseness of such cost data, we feel that the curve represents a tentative upper bound for the expected marginal abatement costs over the next decade.
Huglen and Kvande calculated global mean emissions of about 12 tons of CO2 per ton of primary aluminum including mining, processing of the ore, electrolysis, electricity generation and casting of the metal. [1] Their historical data show a continuously declining trend of specific energy consumption for the electrolytic reduction process, which is consistent with the concept of autonomous energy efficiency improvement (AEEI) used in emission prediction models such as EPPA. For our high, medium and low scenarios we assume that specific CO2 emissions throughout the entire primary aluminum process chain decrease at a rate of 0.5%, 1%, and 1.5% per year, respectively, over the period 1985-2030. As the potential for conversion and retrofitting of old smelters is used up and the energy demand of the aluminum reduction process declines towards its thermodynamic limit, maintaining historical rates of energy efficiency increase will become increasingly difficult. The situation is probably similar for the other parts of the process chain which currently in average contribute less than 25% (3 T per ton of metal) of the overall mean CO2 emissions. [1]
Figure 6 shows the results for the three scenarios on PFCs, CO2 and the sum of both. IPCC estimated that anthropogenic emissions of CO2 were at a level of 26 GT/yr in 1990. [4] Based on the same source we estimate that in 1995 global emissions of greenhouse gases regulated under the Kyoto-Protocol were about 35 GT/yr in terms of CO2 equivalents. The production of primary aluminum was thus responsible for about 0.8% of CO2 emissions and about 1.0% of CO2 equivalent emissions in 1990.
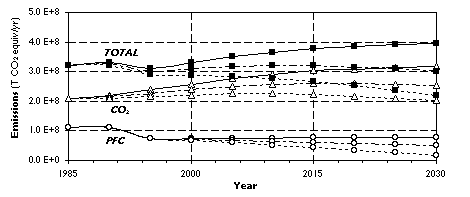
Figure 6. Greenhouse Gas Emissions Scenarios for the Primary A1 Industry.
It is important to keep in mind that decreases in specific energy consumption may be predictable while specific CO2 emissions are not necessarily linked to specific energy demand. If major shifts occur in the composition of energy sources for the electricity being used over the Al production process chain and for the alumina reduction step in particular, the effect on CO2 emissions would be pronounced, as can be seen in Figure 7. For these calculations we assumed that the 1985 power mix of 57% hydroelectricity, 33% coal, 5% nuclear energy, 4% gas and 1% oil would be valid for the old smelters until 2030. [1] We then assumed that additional capacity would be operated with 75% coal and 25% hydroelectric for the high scenario, with the 1985 energy mix for the medium scenario and with 25% coal and 75% hydroelectric for the low scenario. Based on data by Huglen and Kvande, for the EPPA-reference year 1985 the specific CO2 emissions for our three cases would be 18.3, 12.0, 8.8 tons of CO2 per ton of primary metal for the entire process chain. [1]
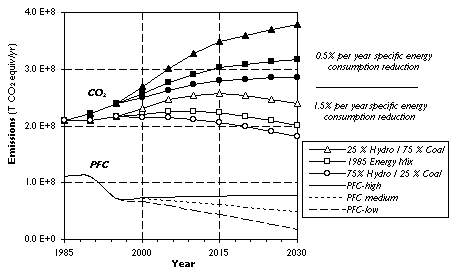
Figure 7. Effect of New Smelter Electricity Sources on Greenhouse Gas Emissions.
We have found that in 1995 about 60% of all emissions of PFCs, 0.8% of all anthropogenic CO2 emissions and 1% of all greenhouse gases regulated under the Kyoto protocol originated from primary aluminum production. The aluminum industry has already succeeded in reducing its global PFC emissions by about 30% since 1990. Our results indicate that further reductions are both a feasible and cost-effective avenue for greenhouse gas mitigation, given current forecasts of the macroeconomic consequences of the Kyoto Protocol. Analysis of the technical potential for abatement suggests that the global primary aluminum industry could achieve a 10% cut in its total greenhouse gas emissions relative to 1995 levels by the year 2010, despite increasing production. Moreover, these reductions can be achieved through quite gradual retrofitting and conversion of older smelting capacity, most of which exists in the Annex B nations. An independent annual assessment of greenhouse gas emissions from the primary aluminum industry should verify continuous progress of abatement efforts.
As a policy question, the process of the aluminum industry's adjustment to the demands of climate change policy does not appear to necessitate government interventions such as the imposition of binding, industry-wide emissions ceilings. Substantial abatement of emissions has been achieved through incremental upgrading of anode technology, instituting automated process control measures, and improving operating practices that prevent the generation of PFCs. The focus of climate change policy at the level of the industry should therefore be on incentives that further encourage producers to undertake such measures.
2. Tabereaux, A.T., 1994, Anode effects, PFCs, global warming, and the aluminum industry, J. of Metals, 30-34.
3. Ravishankara, A.R., S. Solomon, A.A. Turnipseed and R.F.Warren, 1993, Atmospheric lifetimes of long-lived halogenated species, Science, 259:194-199.
4. IPCC, 1996, Summary for policymakers, Climate Change 1995--The science of climate change, Intergovernmental Panel on Climate Change, Working Group 1, Cambridge University Press, Cambridge, UK.
5. Harnisch, J., R. Borchers, P. Fabian, H.W. G%ggeler, U. Schotterer, 1996, Effect of natural tetrafluoromethane (CF4), Nature, 384:32.
6. Harnisch, J., and A. Eisenhauer, 1998, Natural CF4 and SF6 on Earth, Geophys. Res. Lett., 25:2401-2404.
7. Harnisch, J., 1996, Emission scenarios for CF4 and C2F6 derived from reconstructed atmospheric concentrations, Proceedings of Workshop: Successful partnership to reduce PFC emissions from primary aluminum production, May, Washington, DC, Environmental Protection Agency.
8. Zazzera, L., 1995, Comparison of exhaust emissions during C3F8 and C2F6 plasma cleaning of a Novellus Concept One PECVD system using FTIR, 3M Innovation.
9. Japan, 1997, Japan's second national communication under the United Nations Framework Convention on Climate Change, Ministry of Foreign Affairs, Tokyo.
10. USA, 1998, Inventory of United States greenhouse gas emissions and sinks 1990-1996, Environmental Protection Agency, Washington, DC.
11.Harnisch, J., 1997, Die globalen atmosph%rischen Haushalte der Spurengase Tetrafluormethan (CF4) and Hexafluorethan (C2F6), Ph.D-thesis, University of Guttingen, Cuvillier-Verlag, Guttingen.
12. Harnisch, J., 1998, Reactive Fluorine, in: Handbook of Environmental Chemistry--Reactive Halogens in the Atmosphere, P. Fabian and O. Singh (eds.), Springer-Verlag, Berlin, in press.
13. Harnisch, J., R. Borchers, P. Fabian and M. Maiss, 1998, The age of air in the polar middle atmosphere derived from CF4, submitted to Geophys. Res. Lett.
14. Harnisch, J., R. Borchers, P. Fabian, M. Maiss, 1996, Tropospheric trends for CF4 and C2F6 since 1982 derived from SF6 dated stratospheric air, Geophys. Res. Lett., 23:1099-1102.
15.Yang, Z.L., R.S. Eckaus, A.D. Ellerman and H.D. Jacoby, 1996, The MIT Emissions Prediction and Policy Analysis model, Report No. 6, MIT Joint Program on the Science and Policy of Global Change.
16.Ellerman, A.D., and A. Decaux, 1998, Analysis of post-Kyoto CO2 emissions trading using marginal abatement curves, Report No. 40, MIT Joint Program on the Science and Policy of Global Change.
17. UNCTAD, 1984, UNCTAD Commodity Yearbook 1984, United Nations Conference on Trade and Development.
18.UNCTAD, 1994, UNCTAD Commodity Yearbook 1994.
19. Summers, R., and A. Heston, 1991, The Penn World Table (Mark 5): An expanded set of international comparisons, 1950-1988, Quarterly Journal of Economics, 106(2):327-368.
20. Thonstad, J., and A. Oygard, 1995, On the formation of perfluorocarbon gases during anode effect in aluminum cells, Proceedings of the VIII Al Symposium, Slovakia, Ziar Nad Hronom-Dolovany, p. 229-234.
21. Bouzat, G., J.-C. Carraz and M. Meyer, 1996, Measurement of CF4 and C2F6 emissions from prebaked pots, Light Metals 1996, 413-417.
22. Nissen, S.S., and D.R. Sadoway 1997, Perfluorocarbon (PFC) generation in laboratory-scale Aluminum Reduction Cells, Light Metals 1997, 159-164.
23. Leber, B.P., A.T. Tabereaux, J. Marks, B. Lamb, T. Howard, R. Kantemaneni, M. Gibbs, V. Bakshi and E.J. Dolin, 1998, Perfluorocarbon (PFC) generation at primary aluminum smelters, Light Metals 1998, 277-285.
24. Keller, F., and W.K. Fischer, 1992, Anode manufacturing in a changing environment: An overview, Light Metals 1992, 673-686.
25. IPAI, 1996, Anode effect and PFC emission survey 1990-1993, International Primary Aluminium Institute, London.
26. Ghosh, J., A. Steube and B. Levenig, 1997, From 110 to 175 KA: Retrofit of VAW Rheinwerk. Part II: Construction and Operation, Light Metals 1997, 239-242.
27. Grjotheim, K., H. Kvande and Q. Zhuxian, 1995, Key improvements to the Hall-Heroult since the end of World War II, J. of Metals, 47(11):32-35.
28. Kvande, H., 1997, Retrofitting older aluminum reduction cell lines: a way to extend productive life, J. of Metals, 49(2): 21-26.
29. Manaktala, S.C., 1995, The Russian Al industry's difficult transition to a market economy. J. of Metals, 47(5):14-18.
30. Beck, T.R., 1995, A non-consumable metal anode for the production of aluminum with low-temperature fluoride melts, Light Metals 1995, 355-360.
* A previous version of this paper was prepared for the symposium "Abatement of Greenhouse Gas Emissions in the Metallurgical & Materials Process Industry" at the 1999 Minerals, Metals & Materials Society Annual Meeting. This work was supported through a Otto Hahn Fellowship awarded by the Max Planck Society to JH, an award from the MIT Martin Society of Graduate Fellows for Sustainability to ISW, and by the governmental and industrial sponsors of the MIT Joint Program on the Science and Policy of Global Change.
| Top of page |
| |