 |
|
 |
 |
 |
 |
|
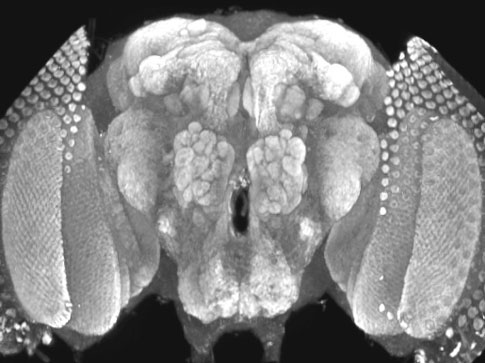
The completion of the genome sequence of Drosophila melanogaster has provided a unique opportunity to initiate a new phase of systematic gene analysis of neuronal function. Although Drosophila as an experimental organism has made fundamental contributions to the fields of heredity and developmental biology, the field of neurobiology has also taken advantage of the powerful genetic techniques afforded by the fruitfly. Seymour Benzer and colleges identified and analyzed mutants with a variety of behavioral defects caused by single gene mutations. Such behavioral mutants initiated a wave of genetic studies into the function of the nervous system and led to the characterization of mutants such as Shibire and Shaker. These studies provided the foundation for a new generation of fly neurobiologists that employed systematic genetic screens for specific neurological phenotypes. The ease of performing electrophysiological analysis at Drosophila neuromuscular junctions readily complements the genetic approaches available in flies, making it an important model system for understanding synaptic function and development, as well as membrane excitability and neurological disease. Using the fruit fly Drosophila as a model, the Littleton laboratory seeks to elucidate the molecular mechanisms underlying synapse formation, function and plasticity by combining molecular biology, protein biochemistry, electrophysiology and imaging approaches with genetics. In addition, studies are underway to characterize how the connections among neurons change during learning and memory. The lab has identified many previously unsuspected gene candidates in the fly brain for activity-dependent modulation of neuronal function and is determining how these genes contribute to cellular forms of behavioral plasticity. In addition, the lab is interested in the alterations in neuron-to-neuron signaling and connections that underlie epilepsy, Huntington’s disease, Autism and other genetically complex neurological disorders. Together, these approaches should greatly expand the understanding of the basic mechanisms of synapse function and plasticity, as well as provide insights into expression changes that allow synaptic ensembles to store information through changes in neuronal connectivity and function. |
|