Why?
Monitoring for levels of dissolved oxygen can be used to indicate the presence of eutrophication in bodies of water. Nitrogen, phosphorus, carbon, and other nutrients are normally the limiting factors in marine ecosystems. Eutrophication occurs when there are excessive nutrient levels in the water; human-induced eutrophication is caused by runoff from chemical fertilizers and animal waste. During eutrophication, a thick layer of algae (such as phytoplankton) covers the surface of the water. Since algae consume more nutrients than other marine organisms, they benefit the most from an increase in nutrient levels. The algae quickly multiply, covering the water's surface and decreasing the amount of sunlight that penetrates the water. As a result, the plants below the water are unable to get the sunlight needed for photosynthesis, and their populations begin to decline. These plants provide food and spawning habitats for many fish, whose numbers decrease as well. Over time, the algae die and sink below the surface, where they decompose and are consumed by bottom-dwellers. This aerobic respiration consumes even more oxygen, and the end result is anaerobic decomposition. As dissolved oxygen levels decrease even more, fish and other aquatic organisms that require aerobic conditions begin to die off (Mid-Atlantic Integrated Assessment 2003). In extreme cases, the demand for oxygen is so high that even the oxygen in sulphates is used up. The leftover sulfur (S2-) is then free to capture more of the remaining oxygen. This sulfur is what causes the rotten egg smell characteristic of eutrophic bodies of water (World Health Organization 2002).
[top]
How do oxygen sensors work?
There are two types of dissolved oxygen sensors: polarographic and galvanic. Both involve the use of a thin organic membrane over an electrolyte and metal electrodes. Oxygen diffuses through the membrane, and the resulting partial pressure is directly proportional to the volume of oxygen flowing through the membrane. The measured oxygen is reduced at the cathode, producing a current that is directly proportional to the concentration of dissolved oxygen. The sensor then converts this information to the proper units of ppm (parts per million) or mg/L (Royce Technologies).
In polarographic sensors, a polarizing voltage applied across the electrodes stimulates the reduction of oxygen, which produces a current. Since the sensors for the Galapagos will be placed directly into the water source and powered by batteries, galvanic dissolved oxygen sensors will be used on the islands. Galvanic sensors require no external voltage, as the oxygen present will spontaneously activate the reaction to produce a corresponding current (Royce Technologies). A typical galvanic sensor will have a KOH electrolyte, a zinc or lead anode, and a cathode made from silver or platinum. Platinum is more costly, but is less susceptible to corrosion. Galvanic probes can last for several months without needing to replace the membrane or the electrolyte (Cole-Parmer Instrument Company, Royce Technologies).
The solubility of oxygen varies inversely with temperature, altitude, and salinity. Many probes can automatically compensate for the conditions at the test site, thereby providing accurate data measurements (Cole-Parmer Instrument Company).
Future Sensors
In March of 2004, In-Situ Inc. released a new kind of sensor called RDO, the Rugged Dissolved Oxygen sensor. This sensor requires no membranes, cleaning, or stirring, and makes use of micro-optical technology. In the presence of oxygen, a red light is emitted, whose intensity varies inversely with the concentration of dissolved oxygen. There is also a large memory for the stored data. The battery life of the long-life lithium battery in each sensor is 58 days (at 20 second intervals) or 14 months (at 20 minutes intervals). Unlike some galvanic sensors, these sensors' readings are not affected by temperature, and they can be submerged up to a depth of 6000m in the ocean (In-Situ.com 2004). However, each sensor costs around $5000, and the technology is relatively new (AquaticLife.ca 2003). It would be more practical to use these sensors in the future, once the price decreases, and once more testing has been done to fix any problems associated with the RDO.
[top]
Where will the sensors be placed?
They will be placed in the streams, lakes, lagoons, and coral reefs of the Galapagos. Many of the streams are close to agricultural zones, and the runoff from fertilizers is a major source of excessive nutrients. The oxygen sensors will be placed at the head of these streams, close to the source of pollution. Part of the Ecovillage's plan is to implement biodynamic farming on the islands, free of commercial pesticides or fertilizers. The oxygen sensors will be able to measure the increase in oxygen levels as these chemicals are phased out. Once the farms become biodynamic, the sensors in the streams can be used to establish a baseline of the streams' natural dissolved oxygen levels. Since the normal range of dissolved oxygen levels is affected by the climate, season, time of day, and the particular organisms in each body of water, such a baseline can only be established through field monitoring. If the oxygen levels are below the normal range, researchers can then go to the site of interest and perform more tests as needed.
Coral reefs are one of the world's most sensitive ecosystems. The dissolved oxygen sensors will be attached by clamps to the floating buoys (another part of the monitoring system) used to monitor the sea and air.
In total, about twenty or twenty-five oxygen probes would be used at a time. Their exact locations will be determined on site, based on the terrain and other practicalities. For example, they should not be placed in areas so isolated that anyone replacing the probes would greatly disturb the local habitat.
This is an example of where the oxygen sensors would be placed. The map depicts a section of San Cristobal; sensors will be placed in El Junco, the only freshwater lake on the islands. Each triangle represents an oxygen probe.
Dissolved Oxygen Sensor Placement
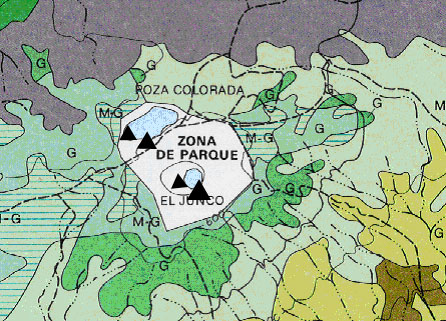
(INGALA: Instituto Nacional Galápagos.)
Below is another example of where the sensors would be placed. The black dots represent coral reefs off the coast of Santa Cruz.
Dissolved Oxygen Sensor Placement
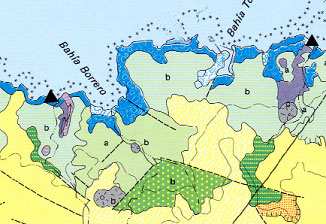
(INGALA: Instituto Nacional Galápagos.)
[top]
Which specific sensors will be used?
There are dozens of galvanic probes available on the market. An example of an appropriate probe is the OxyGuard Model 525, designed for long-term use in the field. It automatically compensates for temperature, and can be solar powered. There are no regular maintenance needs such as frequent calibration. The replacement membranes are screwed on, and anyone can replace them (OxyGuard International 2004). The exact price of the Model 525 is not available, but Neptune Systems has a similar probe, also from OxyGuard, listed as $450 (Neptune Systems). In addition, the cost of a membrane service kit (25 replacement membranes, plus electrolyte fluid) for a similar probe from OxyGuard is $25 (Aquaculture-supply 1999). The replacement time for each membrane will be on the order of several months (like that of galvanic probes in general), and can be better determined by experimentation. Since no experience is necessary, it is possible to hire the locals on the islands to inspect and replace the membranes as needed.
Data should be taken on a regular basis to establish a baseline and also to monitor changes. More frequent measurements should be taken in the beginning, such as once every four hours. Once the data has been examined, a conclusion can be reached about whether or not to increase the time interval.
The data from each sensor can be transferred to a central database by using a radio transmitter. Each transmitter should be solar or battery powered, and small enough to be attached individually to each probe. They also need to be weatherproof, as they will remain at the site under they need to be replaced.
Dissolved Oxygen Sensor

58 mm diameter, 88mm long
OxyGuard Model 525 (http://oxyguard.dk/)
[top]
Sources
- Aquaculture-supply. (1999). New Products Not in Current1998-99 Catalogue. Retrieved November 27, 2004, from
http://www.aquaculture-supply.com/newprod.pdf.
- AquaticLife.ca e-store. (2003). Retrieved November 27, 2004, from http://www.aquaticlife.ca/eStore/shopdisplayproducts.asp?id=554641050&cat=
Sensors+for+all+MP+TROLL+9000+Versions%22.
- Cole-Parmer Instrument Company website. Retrieved November 27, 2004, from
http://www.coleparmer.com/catalog/0304_pdf/A-1110.pdf.
- Elpro Technologies. 505U Radio Telemetry Module. Retrieved November 28, 2004, from http://www.elprotech.com/elpro/505u.htm.
- In-Situ Inc. website. (2004). Retrieved November 27, 2004, from
http://www.in-situ.com/In-Situ/News/2004/RDO/RDO_News.html.
- Mid-Atlantic Integrated Assessment. (2003). Eutrophication. Retrieved November 27, 2004, from US Environmental Protection Agency Web site: http://www.epa.gov/maia/html/eutroph.html.
- Omni-Instruments. Elpro Technologies Fact Sheet. Retrieved November 28, 2004, from http://www.omniinstruments.co.uk/radiotel/elprotech.html.
- OxyGuard International. (2004). OxyGuard Model 525. Retrieved November 28, 2004, from http://www.oxyguard.dk/.
- Neptune Systems. Retrieved November 28, 2004, from http://www.neptunesys.com/priceList.htm.
- Royce Technologies. Polarographic vs. Galvanic Dissolved Oxygen Sensors: Discussion on Differences. Retrieved November 27, 2004, from Technical Bulletin TB02.6: http://roycetechnologies.com/TechBulletins/PartsMillion/TB02~24.PDF.
- World Health Organization Regional Office of Europe. (2002). Eutrophication and Health. Retrieved November 27, 2004, from http://europa.eu.int/comm/environment/water/water-nitrates/eutrophication.pdf.
[top]
Copyright! All rights reserved.
Webmasters


