mini-FastEEM
“Mini-FastEEM”, capable of collecting diffuse white light spectral reflectance and 340 nm-excited fluorescence.
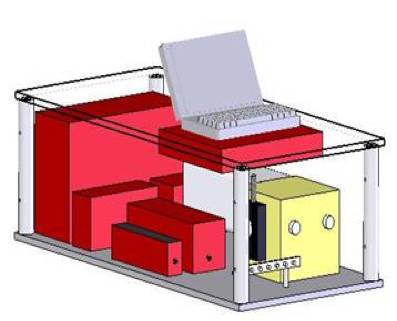 |
Figure 1. Schematic of clinical Fast EEM instrument. |
The DRS/IFS instrument uses two different light sources including a 337 nm N2 laser (NL100, Stanford Research Systems, Sunnyvale, CA) for fluorescence spectroscopy and a Xe lamp (L7684, Hamamatsu Corp., Bridgewater, NJ) for reflectance spectroscopy. The Xe lamp provides a 2.9 µs FWHM pulse of white light, 1 J/pulse max, given an external trigger supplied by the software. The N2 laser provides a 3.5 ns FWHM pulse of 337 nm light, 170 µJ per pulse. Light is delivered and collected from tissue via FastEEM probe and is brought to the entrance slit of the diffraction grating spectrometer (Spectra Pro 150, Acton Research, Acton, MA). The collected light is dispersed onto an intensified CCD detector (PIMAX, Roper Scientific, Princeton, NJ). To insure easy reproducibility, the collection fibers have individual SMA adapters that connect to the spectrograph. The CCD is operated in a gated mode and is thermoelectrically cooled to -20C. The total collection time for fluorescence and reflectance spectra is approximately 0.3 s. Several of these acquisitions can be averaged together to increase the SNR, making a typical acquisition time on the order of 1.5 s. Integrated software (LabVIEW and Matlab) in the system enables rapid collection of DRS and IFS spectra.
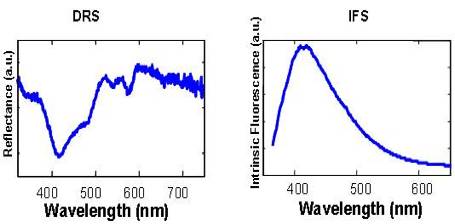 |
Figure 2. Example of DRS and IFS spectra acquired in 1.5s from breast tissue. |
Recent Publications
- Z. Volynskaya, A. S. Haka, K. L. Bechtel, M. Fitzmaurice, R. Shenk, N. Wang, J. Nazemi, R. R. Dasari and M. S. Feld, "Diagnosing breast cancer using diffuse reflectance spectroscopy and intrinsic fluorescence spectroscopy," J Biomed Opt 13 (2), 024012 (2008).
- O. R. Scepanovic, M. Fitzmaurice, J. A. Gardecki, G. O. Angheloiu, S. Awasthi, J. T. Motz, J. R. Kramer, R. R. Dasari and M. S. Feld, "Detection of morphological markers of vulnerable atherosclerotic plaque using multimodal spectroscopy," J Biomed Opt 11 (2), 021007 (2006).
- I. Georgakoudi, B. C. Jacobson, M. G. Muller, E. E. Sheets, K. Badizadegan, D. L. Carr-Locke, C. P. Crum, C. W. Boone, R. R. Dasari, J. Van Dam and M. S. Feld, "NAD(P)H and collagen as in vivo quantitative fluorescent biomarkers of epithelial precancerous changes," Cancer Res 62 (3), 682-687 (2002).


|
 |